|
FAQ Installation Instructions • Upload the plugin files to the /wp-content/plugins/atom-builder directory, or install the plugin through the WordPress plugins screen directly. • Activate the plugin through the ‘Plugins’ screen in WordPress • That’s it! Edit your pages in the customizer and add widgets to your page to see it in action! Does the Atom Builder supports custom post types? By default, it only works with basic pages. But there’s a hook for that! Developper documentation is in writing, don’t worry. Have a look in the init-functions.php file in the inc/ folder to see how it works. Do you plan on releasing other widgets? 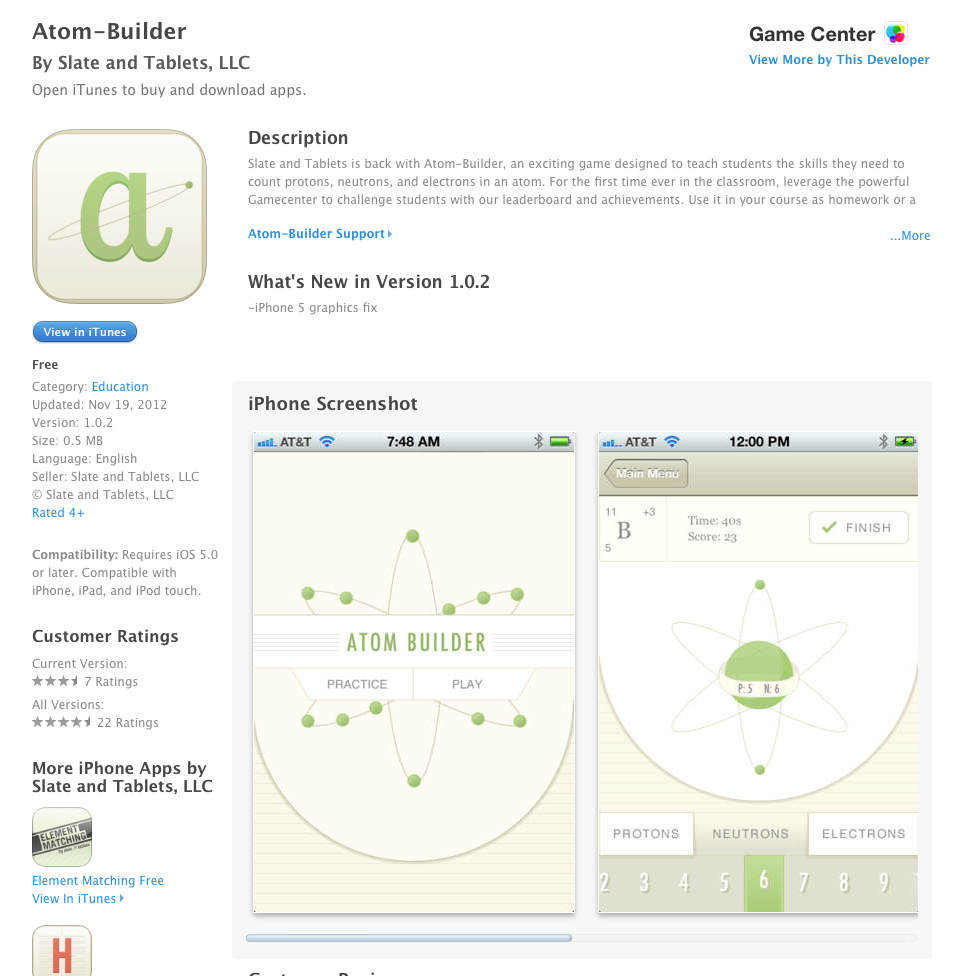  
The three widgets included are just a start. Phet.colorado.edu Build an Atom - Atoms| Atomic Structure| Isotope Symbols - PhET. 600 × 394 - 87k - png pbs.org A Science Odyssey: Atom Builder: Building an Atom 216 × 171 - 5k - gif pbs.org A Science Odyssey: Atom Builder: Description 441 × 304 - 8k - gif pbslearningmedia.org Atom Builder| Science| Interactive| PBS LearningMedia 710 × 399 - 47k - jpg xula.edu General Chemistry 600 × 394 - 81k - png usarmystars.com Stars 2732 × 2048 - 634k - png rayslearning.com Screenshot of Atoms, Bonding and Structure - Interactive Chemistry. 643 × 502 - 17k - gif phet.colorado.edu Build an Atom - Atomic Structure| Atoms - PhET Interactive. 300 × 226 - 25k - png students.ga.desire2lea. Physical Science 400 × 309 - 99k - png orau.org Build An Atom 750 × 500 - 236k - png sciencenetlinks.com Atom Builder - Science NetLinks 225 × 165 - 13k - jpg chemedx.org Building Atoms: One App at a Time| Chemical Education Xchange 2048 × 1536 - 2028k - png phet.colorado.edu Isotopes and Atomic Mass - Isotopes| Atomic Mass - PhET. Create a file called.atom-build.yml (note the inital dot). Build an Atom Atom Symbol Game. Build an Atom Atom Symbol Game. Atom Builder. Building a carbon atom from scratch, using subatomic particles as building blocks, is simply not something humans can do. Even with the most powerful. 600 × 394 - 90k - png sliderbase.com Protons (+) - SliderBase 960 × 720 - 192k - png blog.fortinet.com From Shark to Atom: Ransomware Service Offers Generous Returns. 752 × 374 - 9k - png catalog.keep.edu.hk Atom- Builder - KEEPCatalog 636 × 460 - 47k - jpg slideplayer.com Bohr”ing you with atoms The study of chemistry begins with the. 960 × 720 - 62k - jpg olliehooper.com Atom Builder – Olliehooper 960 × 541 - 199k - jpg zitogiuseppe.com Leonardo: Interactive Virtual Science Museum 416 × 278 - 7k - gif ansto.gov.au Games - ANSTO 800 × 517 - 70k - jpg. Matter is all the 'stuff' that we see, feel, and smell around us. By definition, matter has mass and takes up space. It includes substances like water, wood, rock, metal, plastic, air, and countless other materials. All types of matter are built from tiny particles called atoms. Atoms and the subatomic particles they're made of are far too small to be seen, even with the most powerful microscopes. Despite this, physicists have developed an understanding of their structure using experimentation and indirect observation. Models of the structure of atoms have changed a great deal since the first -- a simple, undifferentiated sphere -- was proposed in the nineteenth century. In today's atomic model, a 'cloud' of orbiting, negatively charged particles called electrons surrounds a small, dense nucleus of positively charged protons and neutral neutrons. For many decades, physicists thought that protons, neutrons, and electrons were fundamental, which means structureless, or not made of anything smaller. While it appears that electrons are, in fact, fundamental, physicists now know that protons and neutrons, the particles that make up an atom's nucleus, are built from even smaller particles called quarks. Scientists have identified six known quarks, which combine in groups of three to create either positively charged protons or neutral neutrons, depending on their combination. Most elements have both a stable form and one or more unstable forms. An element is stable when its subatomic particles are in a particular numerical balance. 
For example, a carbon nucleus with six neutrons and six protons is stable, meaning that it is unlikely to give off a particle to improve its stability. An atom with too many or too few neutrons in relation to its number of protons will be radioactive, meaning that it is likely to undergo radioactive decay, a process in which the nucleus gives off high-energy particles. Sometimes a radioactive element gives off a proton, changing its elemental identity in the process.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
May 2019
Categories |
 RSS Feed
RSS Feed
